Density Curriculum
Section 3—Lesson 9: How Do Atomic and Molecular Bonds Contribute to Density?
Lesson Plan
Materials
- Cause #1 and Cause #2 posters (PDFs)
- Two sets of balls with springs between them to illustrate atomic bonds (see picture below)
- Water Molecule illustration (PDF 162 KB)
- Molecular Diagram of Graphite (PDF 226 KB)
- Molecular Diagram of Diamond (PDF 247 KB)
- How and Why article by David Ropeik (PDF 96 KB)
- The Densest Element sheet (PDF 69 KB)
- Two of the cylinders from Lesson 1
Prep Step
- Review the lesson plan, background information, and understanding goals.
- Gather materials.
- Enlarge the Cause #2 poster to post beside the Cause #1 poster.
- Photocopy the article How and Why by David Ropeik and the sheet, The Densest Element.
Analyze Thinking
Step 1: What Else Causes Differences in Density?
Remind students of the discrepancies between what atomic mass predicts for the density of a substance and the information from the density chart. In most cases, high atomic mass correlates with high density, but not in all cases. Ask the students to consider what else might be going on. Do they have any ideas? Collect their thoughts.
Remind them of the various models that they considered in Lesson 2. Do those models help them to think about what else might be going on?
Explore Causality
Step 2: Introducing the Role of Atomic and Molecular Bonds
Put up the Cause #2 poster. (Leave the poster from the last lesson up beside it for comparison.) Explain, "In order to consider atomic mass, we zoomed in to the micro-level. Now we are going to stay zoomed in at that level because Cause #2 is also at the micro-level. A second cause of density has to do with the way that the atoms stay together or that collections of atoms are structured."

Show the students a set of two balls with a spring between them. Explain that each ball represents an atom (along with its protons, neutrons, and electrons) and the spring represents the bond between them. Then compare it to another set of two balls with a shorter spring between them. Show how it is harder to pull the stronger (shorter) bond apart so the two atoms stay closer together. The weaker (longer) bond is easier to pull apart, so the two atoms can be further apart. The strength of the bonds affects how close the atoms are to each other, so it affects how many atoms fit in a certain amount of space. Stronger bonds contribute to greater density. Make sure that the students realize that the bonds are not actually matter and that the springs are there to symbolize electrical attraction.
Explain that atoms can combine to make compounds and molecules. Atoms may be bonded into molecules or compounds by atomic bonds, and molecules are bonded to other molecules by molecular bonds.
Next, draw their attention to how atomic bonds affect the structure of how atoms come together into molecules. The way that atoms come together into molecules depends upon how they organize themselves, and this is governed by negative and positive charge. The negatively charged atoms and the positively charged atoms attract each other, while negatives repel other negatives and positives repel other positives.

Draw a water molecule on the board to show how the atoms connect to form a molecule that looks like mouse ears (see picture to left and Water Molecule illustration). A second cause of density has to do with the amount of space between the atoms due to the strength and the structure of their atomic bonds (the bonds between atoms).
Show students the Molecular Diagram of Graphite and the Molecular Diagram of Diamond. Each one is a compound made of carbon atoms. How they are bonded makes the difference between having graphite or diamond, and makes a very big difference in density!
Graphite ©www.reciprocalnet.org |
Diamond ©www.reciprocalnet.org |
Explain that the structure and spacing of the atomic bonds (in molecules and in compounds) as well as the molecular bonds (between molecules) affect the density of a substance.
Step 3: Is There Air in Between the Bonds?
Ask the students to think about the following question: "Is there air in between the atomic bonds?"
Have students present arguments for and against and explain their reasoning.

Explain that most gases are molecules with at least two atoms. Show a model of a molecule from the air. This molecule would not fit in the space between average atoms in a molecule (see picture to right) which can be measured by the average bond length. Even in the case of an atom that travels alone as a gas (for instance, helium) there isn't really enough room. When atoms bond together, they actually fit more closely than when they just pack together because they are sharing electrons. There are more electrons between atoms that are just packed together but not bonded, and these electrons take up space. The models of bonds that we use make it hard to realize this. It is one way that the models don't work so well.
Beyond this, there are particular reasons why atoms bond or push away from each other. So there are other reasons, besides size, why the spaces between the atoms are not filled with molecules or single atoms that make up the air.
Again, stress that the empty space between the atoms IS NOT air.
Remind the students that the structure and spacing of the molecular bonds (as opposed to the atomic bonds) also affects the density of a substance. We need to zoom out a little bit to consider the molecular bonds. Could the molecules and atoms that make up the air fit in the spaces in between molecular bonds? Gather the students' ideas. In most cases, the answer is no. However, in some cases, the answer is yes. It depends upon the specific molecules and atoms in question. (For instance, in some plastics, gases can fit between the molecules because of the curly structure of the molecules.)
Review, Extend, Apply
Step 4: When One Cause Overrides Another Cause
Come back to the gold and platinum puzzler from Lesson 8, Step 6. Do students have any ideas about what could be going on now? Gather their ideas. Have students read the article How and Why by David Ropeik and answer the questions about it on the sheet, The Densest Element sheet.
Step 5: Revisiting the Aluminum and Copper Cylinders From Lesson 1
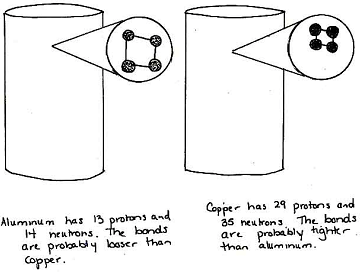
Show the students the cylinders from Lesson 1. Ask them to revisit the models that they drew in that lesson. They should draw the cylinders and show a microscopic slice of the cylinder—what they would see if they could look under the microscope (see example below).
Student Example: Microscopic View of Cylinder
This student depicts how atomic mass (Cause #1) and the bonds (Cause #2) might affect the density of the aluminum and the copper. The student knows that copper has more atomic mass than aluminum and thinks that the bonds might also be tighter, contributing to overall higher density. Notice that, in this case, the student attends to the length of the bonds but not the structure of the bonding. As students learn more about the nature of atomic and molecular bonding, they may begin to incorporate this information into their models as well.